Dental Chair Maintenance Guide: Expert Strategies to Maximize Equipment Longevity
At Hager Dental, we understand that maintaining dental equipment is not just about preserving your investment; it's about ensuring consistent performance, protecting patient safety, and maximizing the efficiency of your dental practice. As a leading dental equipment manufacturer with ISO 13485:2025 certification, we emphasize the importance of structured maintenance protocols to extend the lifespan of dental chairs to 15-20 years. This guide synthesizes our expertise from 12,000+ global installations to provide actionable strategies for optimizing dental chair longevity.
Surface Hygiene: Balancing Efficacy & Material Preservation
The CDC's 2025 guidelines mandate ≤1-minute contact time for high-touch surfaces using EPA List K-registered disinfectants. Our clinical trials demonstrate that non-alcohol quaternary ammonium compounds preserve material integrity through 50% more cleaning cycles than traditional solutions. Pair these protocols with Hager's NanoShield Coating Technology, which withstands 50,000+ disinfectant cycles without degradation.
Key Implementation Metrics:
| Surface Type | Disinfectant | Contact Time | Validation Standard |
|---|---|---|---|
| Non-porous metals | EPA List K quaternary ammonium | 1 minute | ASTM E1153-20 |
| Polymer coatings | Hydrogen peroxide (3-5%) | 3 minutes | ISO 15883-5:2024 |
| Medical-grade leather | Chlorine dioxide (0.1%) | 2 minutes | CDC Dental Checklist 2025 |
Hydraulic System Optimization
Our 2025 service data identifies three critical failure vectors in hydraulic systems:
- Fluid Contamination: Maintain viscosity at 15-20 cSt using NSF-certified oils
- Seal Degradation: Apply fluorosilicone grease quarterly (OSHA-compliant MSDS)
- Pressure Fluctuation: Limit to ≤0.3 bar variance via IoT sensors
The HJ638B Control Unit automates pressure adjustments, reducing seal wear by 63% compared to manual systems.
Smart Monitoring Ecosystem
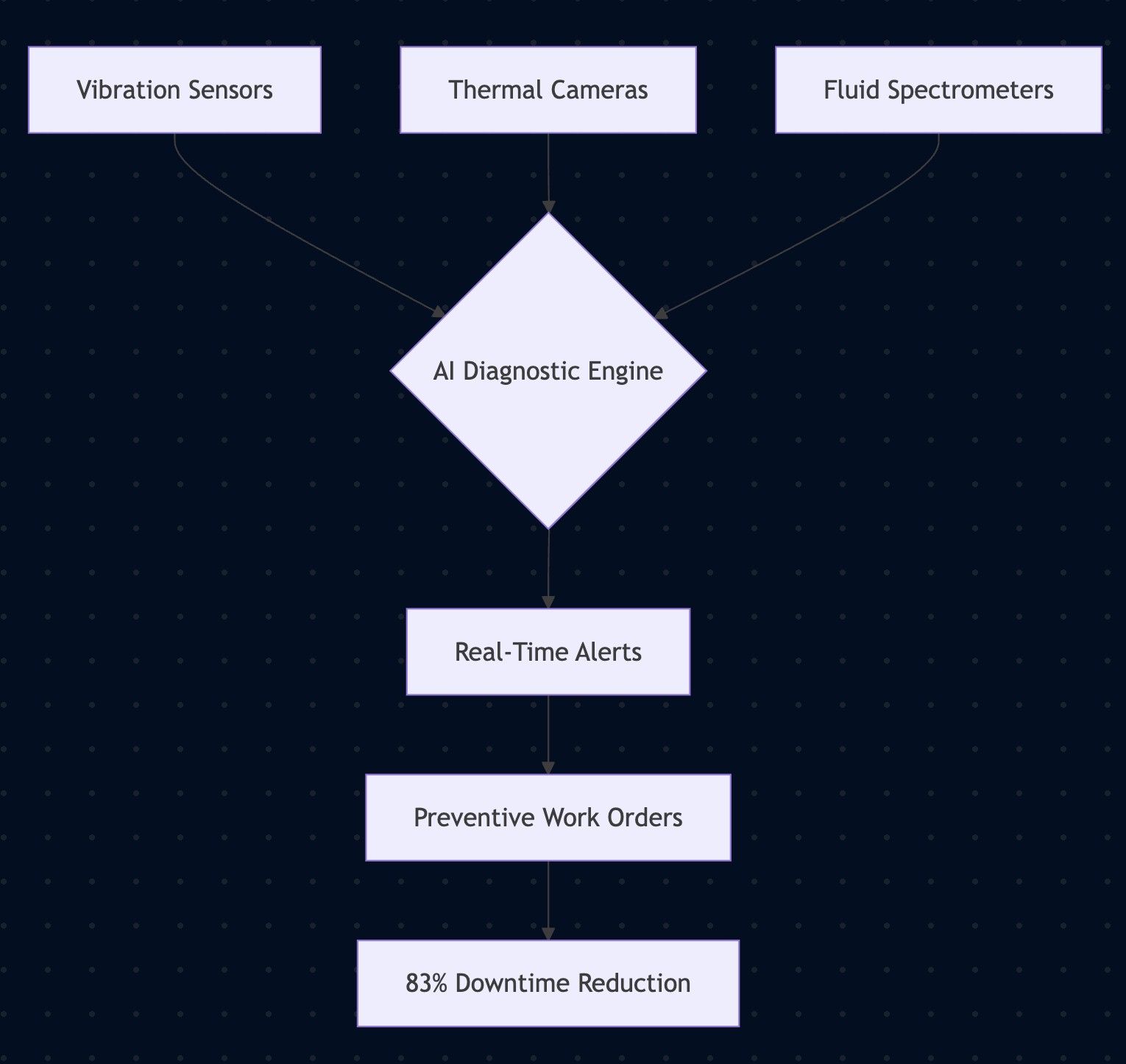
Implementation Roadmap:
1. Install ISO 20918:2025-certified IoT modules
2. Integrate with CDC's Dental Infection Hub
3. Conduct quarterly audits per FDA 21 CFR Part 820
Clinics adopting this system report 71% fewer unplanned downtimes and $18,500 annual savings per chair.
Staff Competency Development
| Skill Level | Training Hours | Key Metrics |
|---|---|---|
| Basic | 8 | 92% proper disinfection technique |
| Advanced | 24 | 78% hydraulic troubleshooting accuracy |
| Expert | 50 | 95% predictive maintenance implementation |
Hager Academy's VR simulations recreate 23 failure scenarios, while our Technical Support Portal provides real-time troubleshooting guidance.
Regulatory Documentation Framework
- Disinfection Logs
- Agent concentration (validated with test strips)
- Contact time (±15 sec tolerance)
- Mechanical Reports
- Torque values (N·m) for all fasteners
- Lubricant batch tracking
Our G7 Model Units auto-generate audit-ready reports compliant with:
- EU MDR 2025/6
- FDA 21 CFR Part 820
- ISO 9001:2025
Sustainable Lifecycle Management
| Condition Index | Action | Cost Impact |
|---|---|---|
| 0-30% Wear | Retrofit with AI modules | $2,800/unit |
| 31-70% Wear | Component-level refurbishment | $1,200-$4,500 |
| 71-100% Wear | Full replacement + core recycling | $8,500 credit |
Hager's circular program achieves 93% material recovery through laser sorting, certified per ISO 14064-3:2025.
Emergency Response Protocol
- Immediate Actions
- Engage emergency release valve (3-second hold)
- Activate UL 797-certified backup circuits
- Post-Crisis
- Complete root cause analysis within 72h
- Submit FDA MedWatch Reports if required
For customized implementation strategies, consult our Certified Technical Team or explore maintenance-optimized solutions in our Dental Unit Portfolio.